
制药与生物科技板块能否延续近期上涨势头?受多年利空因素拖累后,得益于政策风险更趋明朗化及并购交易回暖趋势的提振,去年底这些板块的市场情绪转趋积极。
尽管外界对美国食品药品监督管理局(FDA)的领导层更迭和资金限制问题存在担忧,但该机构整体运作如常,2025年的获批药物数量与长期平均水平基本持平。随着贸易政策和药品定价改革逐渐明晰,越来越多的投资者将其潜在影响视为可控,从而推动制药与生物科技板块去年强劲反弹。
这一强劲表现一直延续至2026年。除利空因素得以缓解外,在人工智能驱动的颠覆性变革引发不确定性,令市场其他板块承压的情况下,制药与生物科技板块的防御性特征或也为其提供了额外支撑。与此同时,市场广泛认同人工智能可以赋能药物研发能力,有助于加速研发进程并缩短上市周期。
自2025年中以来,制药与生物科技板块表现显著优于大盘
价格表现(2025年6月30日至2026年2月24日)。
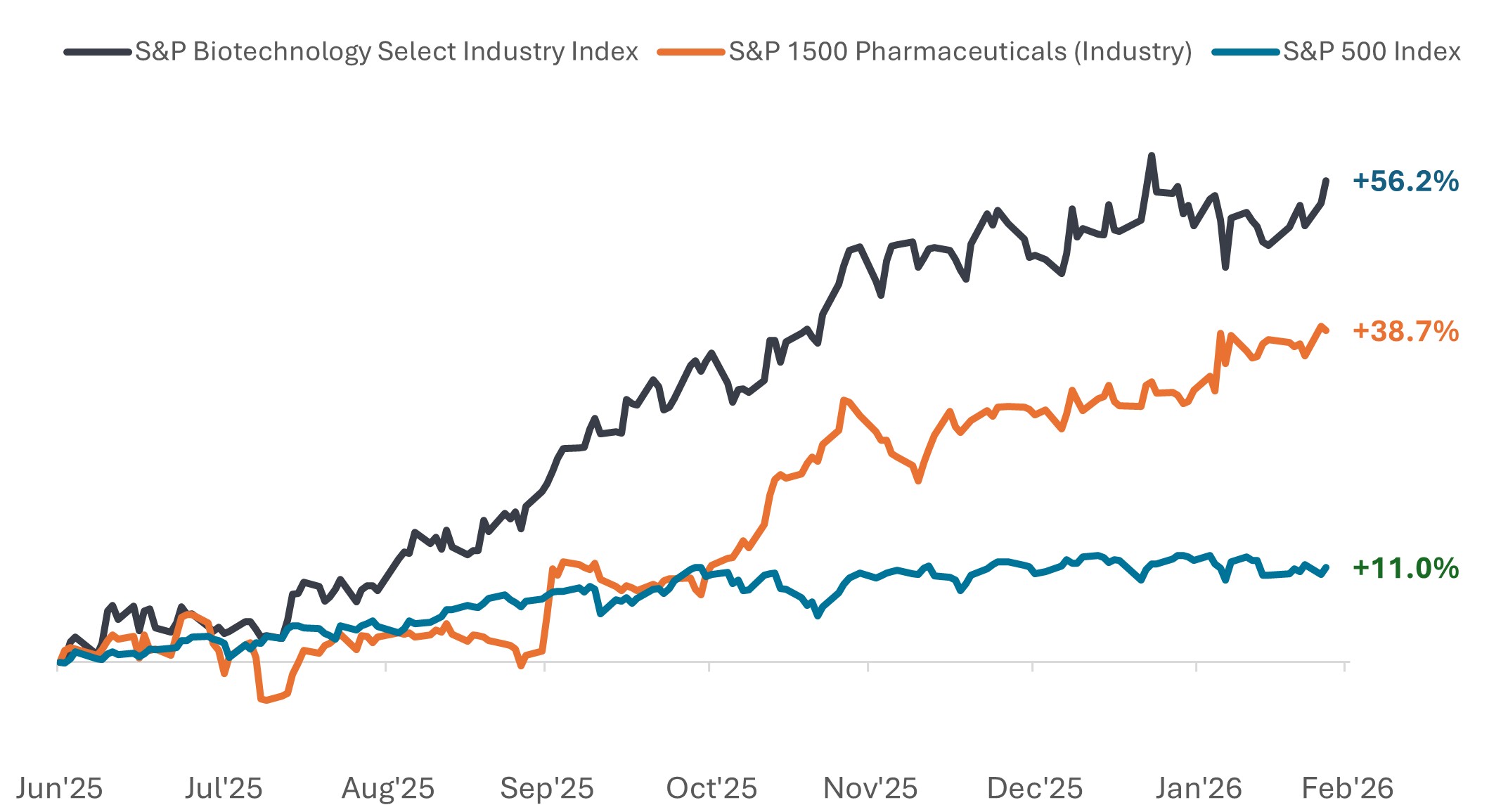
资料来源:彭博,数据涵盖2025年6月30日至2026年2月24日。标普生物科技精选行业指数包括标普总市场指数中被归类为GICS生物科技子行业的成份股。标普1500制药(行业)指数包括纳入标普综合1500®指数,并归类为GICS制药行业的成分股。过往表现并非未来业绩的预测。
在更为积极的环境下,2026年行业有望迎来众多潜在催化因素
透明度提升与市场表现强劲共同营造了更为有利的环境,而围绕战略并购交易回暖的乐观情绪重燃以及首次公开募股(IPO)市场可能重启的预期,则进一步巩固了这一态势。更为重要的是,日益向好的宏观环境已令市场关注点重新回归至长期以来驱动该行业的核心要素:创新。
在此方面,2026年有望成为催化剂密集之年,多个主要治疗领域将迎来一系列备受期待的新药上市、临床试验结果及监管决策的发布。在下文中,我们将重点梳理投资者应密切关注的部分重要事件,以寻找可能塑造未来一年行业动态的信号。
大型制药公司:主流治疗模正处于变革之中
对制药行业而言,数个备受瞩目的临床及监管里程碑事件将检验近期的创新能否转化为持久的增长。新的治疗模式也在不断涌现。后续一系列事件有可能重塑现有标准疗法,并影响投资者对行业竞争格局的评估。
Obesity & cardiometabolic treatments: From proof‑of‑concept to scale and competition
经过多年的临床验证与市场检验,可以明确的是,肥胖症市场对GLP-1疗法有效性的质疑已成为过去。2026年的关注点将转向该等药物的给药方式、规模化应用广度,以及患者和消费者将如何在不同的治疗方案中作出选择。投资者还将关注,随着更多支付渠道的打通,药物的可及性、可负担性以及市场的长期规模将受到怎样的影响。
值得关注的催化因素:
- Oral GLP‑1s (“year of the pill”): Real‑world uptake of oral formulations – most notably Novo Nordisk’s recently approved oral Wegovy (semaglutide) and the pending FDA decision on Eli Lilly’s orforglipron (expected April 2026) – will test whether pills can meaningfully expand the treated population beyond injectables. Importantly, oral therapies could broaden global reach by simplifying distribution and storage, potentially improving access in markets where injectable cold‑chain logistics are a constraint.
- Next-generation combinations: Late‑February Phase 3 data showed Novo Nordisk’s experimental shot CagriSema delivered strong weight loss but fell short of Eli Lilly’s high-dose Zepbound in a head-to-head trial. Patients on CagriSema lost 23.0% of body weight over 84 weeks, compared with 25.5% for Zepbound, underscoring the high efficacy bar for GLP-1 combinations.
- Multi-agonists: Ongoing late‑stage readouts from Eli Lilly’s retatrutide injectable throughout 2026 will help determine whether activating multiple metabolic pathways can push weight‑loss results meaningfully higher without creating side‑effect tradeoffs that limit long‑term use. For investors, the balance between added efficacy and patient tolerability is critical as companies look for the next step beyond today’s GLP‑ Competition is also building, with Amgen’s monthly MariTide, Roche’s CT‑388, and Boehringer’s survodutide advancing in late‑stage trials, alongside a growing wave of amylin‑based therapies entering or approaching Phase 3, led by Novo Nordisk, Eli Lilly, and Roche.
神经科学:推动治疗更早、更广泛地开展
神经科学正成为又一个有望通过创新显著扩大治疗人群的领域。试验设计与生物标志物的进步,使得研发项目能够在疾病进展的更早期阶段介入,并探索超越传统疗法的新机制。
值得关注的催化因素
- Earlier‑intervention Alzheimer’s trials: Investor attention is increasingly focused on studies evaluating treatment at earlier stages, before symptoms become apparent but when biological signs of Alzheimer’s are already present. Eli Lilly’s TRAILBLAZER‑ALZ 3 trial (TB3) of donanemab in biomarker‑defined, preclinical Alzheimer’s disease is widely viewed as a potential category‑shaping program. While full results are not expected until 2027 and interim disclosure in 2026 is uncertain, any update could influence sentiment around earlier intervention and long‑term market expansion opportunities.
- Next-generation delivery technologies: Newer amyloid programs are focused on improving how treatments reach the brain to enhance safety and patient convenience. Brain‑shuttle technologies, including programs in development from Roche and private companies such as Korsana, aim to deliver higher drug exposure while potentially reducing side‑effect risk and enabling less frequent and subcutaneous dosing. If successful, these advances could address key limitations of earlier therapies and support broader adoption.
- Symptom‑focused programs: Several late‑stage programs are targeting neuropsychiatric symptoms such as psychosis and agitation, where unmet need remains high and clinical endpoints are more straightforward. In Alzheimer’s psychosis, Bristol Myers Squibb is expected to report multiple ADEPT readouts for Cobenfy in the second half of 2026, while Acadia and Alkermes anticipate mid‑ to late‑2026 data from Phase 2/3 programs. In agitation, Axsome is also awaiting a potential FDA decision for AXS-05 in mid‑ For investors, these programs stand out because clearer symptom improvement can translate more directly into clinical adoption and commercial uptake if approved.
- Mechanistic diversification: Beyond treatments that target amyloid plaques, investors are increasingly watching programs aimed at other underlying drivers of neurodegeneration, including tau pathology, inflammation, and synaptic function, which are linked to how brain cells communicate and deteriorate over time. Readouts from programs at Biogen, Eisai, Acumen, and ProMIS Neurosciences through 2026 will help inform whether these alternative approaches can complement first‑generation therapies or extend benefits to a broader group of patients.
生物科技:多重催化因素即将涌现
在生物科技领域,2026年将迎来一系列多元化催化因素,在肝脏疾病、肾脏疾病、肿瘤学以及众多罕见病和专科适应症方面,将有密集的数据和监管决策发布。随着研发管线的推进,这些事件可能共同推动显著的分化趋势。
值得关注的催化因素:
- 肝脏/MASH关键数据:2026年最受关注的生物科技事件之一是Inventiva公司针对lanifibranor治疗代谢功能障碍相关脂肪性肝炎(MASH)的3期NATiV3试验。MASH是与肥胖和糖尿病相关的一种严重的脂肪性肝病,当前治疗手段有限。鉴于该领域之前的后期项目结果喜忧参半,预计在今年下半年公布的顶线结果将具有重大意义。在这个当前商业预期仍较为谨慎的领域中,积极的试验结果或有助于重建市场对靶向多疾病通路的口服疗法的信心。
- Renal regulatory milestones: Several high‑visibility FDA decisions are expected in kidney disease in 2026, led by Travere Therapeutics’ FILSPARI, which is seeking a label expansion into focal segmental glomerulosclerosis (FSGS), a rare and progressive disorder that can lead to kidney failure and currently has limited treatment options. The FDA extended its decision date to April, making this one of the most closely watched renal regulatory events of the year. A positive decision would represent a meaningful clinical advance in a small but commercially important rare‑disease market.
- Oncology moving toward biology‑driven programs: In cancer care, innovation is increasingly shifting toward treatments tailored to the underlying biology of a tumor, with the goal of better matching drugs to the patients most likely to benefit. Several high‑profile readouts in 2026 will test whether this approach can deliver meaningful differentiation. Notably, Roche’s giradestrant data in first‑line metastatic breast cancer, expected in late March or early April, will serve as an important read‑through for Olema’s palazestrant (with Phase 3 OPERA‑01 data expected in the second half of the year) and AstraZeneca’s camizestrant in the SERENA‑4 Other influential readouts include Ideaya’s darovasertib‑plus‑crizotinib combination in metastatic uveal melanoma, with Phase 2/3 data anticipated in late March, and Revolution Medicines’ Phase 3 study in second‑line pancreatic cancer, with results expected in the first half of 2026. Together, these readouts will help clarify whether more targeted approaches can deliver meaningful differentiation in competitive settings.
Implications for investors
随着2025年初的政策不确定性成为过去,2026年制药与生物科技板块的表现将越来越多地由基本面因素驱动:即企业执行力、临床里程碑事件以及研究管线推进。
在重磅药物上市、数据及监管决策密集发布的环境下,投资者有充分的理由在全年中对该板块保持关注。对于那些着眼于长远的投资者而言,从短期市场纷扰中辨别出持久的创新将是关键所在。
倘若英文版本与中文版本出现歧异,概以英文版为准。
S&P 500® Index reflects U.S. large-cap equity performance and represents broad U.S. equity market performance.
标普生物科技精选行业指数包括标普总市场指数中被归类为GICS®生物科技子行业的成份股。
标普1500制药(行业)指数包括纳入标普综合1500®指数,并归类为GICS® 制药行业的成分股。
重要资讯
Diversification neither assures a profit nor eliminates the risk of experiencing investment losses.
Equity securities are subject to risks including market risk. Returns will fluctuate in response to issuer, political and economic developments.
健康护理产业受制于政府监管和报销率以及当局对产品和服务的审批,这些因素可能对价格和供应造成重大影响,亦可能受到迅速过时和专利到期的重大影响。
首次公开募股(IPO) 属于高度投机性投资,可能面临流动性较低及波动性较大的风险。与IPO相关的特殊风险包括经营历史有限、交易历史尚浅、高换手率以及业绩不可复制性。